Functional Aging and Hormone Health
What role does the endocrine system play in healthy aging and the optimization of exercise programming?

In the past decade or so, the word functional has become a fitness industry buzzword, often referring to training systems and movements that take a different or unique approach, but not describing the approach with much specificity. Although there may be gray areas when it comes to defining functional training and functional aging, most fitness professionals can agree that it focuses on enhancing natural movement through all planes of motion, thereby improving performance efficiency in everyday activities.
This is of particular importance when working with older adults, since aging is accompanied by a progressive decline in multiple physiological functions, including the ability to perform daily activities. The topic of “functional aging” addresses this ability (or lack thereof) and is becoming more and more relevant, given the increase in people 65 and older and the disability rates in this population.
According to the U.S. Census Bureau, 14.9% (47.8 million) of adults were 65 years and older in 2015, an increase of 1.6 million from 2014. This number is expected to rise to 98.2 million by 2060 (U.S. Census Bureau 2017). Fitness professionals have a huge opportunity to cater to the needs of this growing population by providing programs that enhance functional-aging factors. To do so, it’s important to understand related biological mechanisms; specifically, the role of hormones in association with aging and the accompanying decline in function.
What Is Functional Aging?
Before diving deeper into the concept of functional aging, let’s define aging. According to Zjacic-Rotkvic, Kavur & Cigrovski-Berkovic (2010), there are five basic descriptors:
- cumulative—the effects of aging increase over time;
- universal—all members of a species show the effects of aging;
- intrinsic—changes occur regardless of external factors;
- progressive—aging involves a series of gradual changes; and
- hurtful—normal function is compromised.
The most common references to aging use chronological terms. Statistics and reports typically categorize people by age rather than by health status or by how well they can perform daily tasks and be active. The latter is the measurement of functional aging, which “rests on the premise that a measure other than chronological age could better reflect one’s position in the ageing process” (Guralnik & Melzer 2002). Functional aging may be a better assessment of “the whole older person.”
While Zjacic-Rotkvic, Kavur & Cigrovski-Berkovic’s definition provides an overarching view of aging, Gault & Willems (2013) focus on more specific outcomes, defining aging as a “multi-factorial process that ultimately induces a decline in our physiological functioning, causing a decreased health-span, quality of life and independence for older adults.” All three of these, but especially the decline in physiological functioning and independence, are central concerns for functional aging. When an individual is no longer able to perform basic, everyday activities such as rising from a chair, climbing stairs, reaching overhead, or carrying and handling small objects, his or her functional ability is greatly affected.
Kalapotharakos et al. (2005) state that “reduction of muscle strength in lower limbs is related to the loss of functional capacity and increased number of falls in older adults.” This functional decline is often led by age-related loss of muscle mass, known as sarcopenia, starting after age 35. This loss progresses at an average annual rate of 1%–2% until age 60 and accelerates to 3% per year after that (Vitale, Cesari & Mari 2016). Even more detrimental to functional ability than sarcopenia is loss of muscle strength, which declines as much as 50% with increasing age (Kalapotharakos et al. 2005). Both skeletal muscle mass and function are influenced by chronic inflammation, which is a strong predictor of disability and frailty in older adults (Nicklas & Brinkley 2009). Although many factors play a role in aging, one important key in the decline of muscle mass and strength—as well as the overall decline in functional ability and the rise in chronic inflammation—is a complex set of changes within the endocrine system.
See also: Pillars of Functional Training for Active Aging
The Role of the Endocrine System
The endocrine system’s pivotal role in aging was first introduced by Vladimir Dilman when he developed the neuroendocrine theory of aging in 1954 (Dean 2018). His theory describes the central role of the hypothalamus—its loss of receptor sensitivity and subsequent loss of homeostasis—in aging. This loss of hypothalamic sensitivity alters hormone concentration, reduces the number of neurotransmitters and can lead to the collapse of metabolic regulatory functions. For example, glucose tolerance may decrease and HDL cholesterol levels may drop, both of which accelerate functional decline and aging (Zjacic-Rotkvic, Kavur & Cigrovski-Berkovic 2010; Kim & Ju 2017).
It is beyond the scope of this article to go into depth on the endocrine system; however, to better understand the role of hormones in functional aging, it’s important to have some rudimentary knowledge of the connections among the main factors. The summary that follows provides the most relevant highlights.
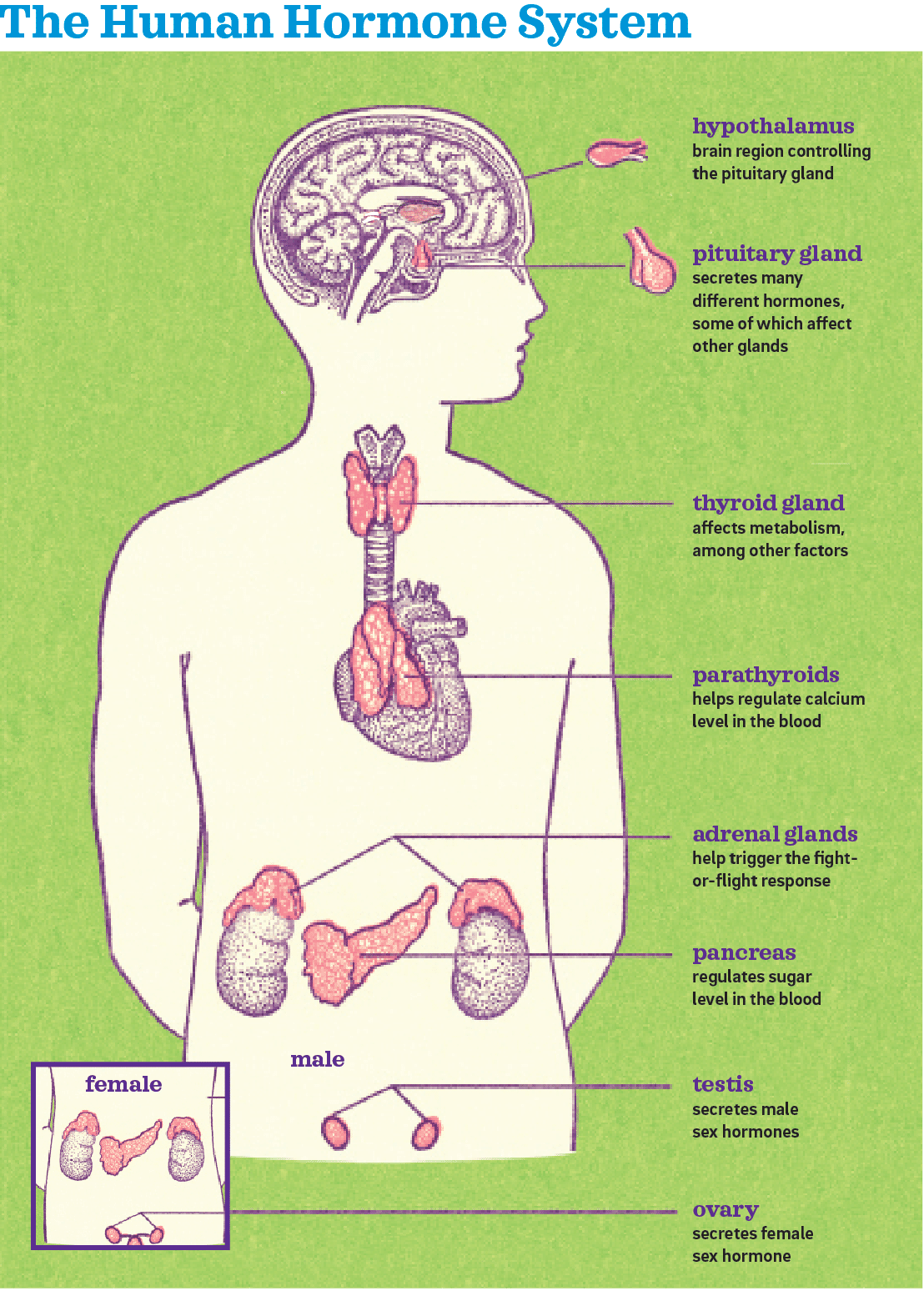
In the brain, two main structures play a role: the hypothalamus and the pituitary gland. “Releasing hormones” from the hypothalamus are sent to the pituitary gland, which in turn sends “stimulating hormones” to other glands—such as the adrenal, thyroid and reproductive organs—to activate the specific hormones required for a desired function
(see “The Human Hormone System,” below).
The interplay of the hypothalamus, pituitary and adrenal glands is of particular importance to aging. This interplay is referred to as the hypothalamic-pituitary-adrenal (HPA) axis, and its main function involves stress response (Ferrari et al. 2001; Ebner et al. 2015; Heaney, Phillips & Carroll 2012). The HPA plays a fundamental role in the interactions among the endocrine, nervous and immune systems and is one of the most adaptive systems (Ferrari et al. 2001). When active, it allows the body to respond effectively to challenges and stress, and when inactive, it can lead to stress response overexposure (Ferrari et al. 2001; Vitale, Cesari & Mari 2016). As a person ages, the homeostatic balance and ability of the HPA axis to respond to stimulation is disrupted by changing hormone levels and by the shift in interactions among hormones. This results in chronic glucocorticoid exposure, higher oxidative stress and increased inflammation (Archer et al. 2011).
Age-related endocrine changes influence every part of the body in some way, and the changes are not attributable to the actions of a single hormone, but rather to the coaction of many hormones throughout multiple systems (Ebner et al. 2015). The hormones that play a part in functional aging are too many to list, but these are of particular interest: dehydroepiandrosterone (DHEA), cortisol, estrogen, testosterone, growth hormone and insulin-like growth factor type 1.
Hormones: An Overview
DHEA
DHEA is produced by the adrenal gland and is the “mother hormone” to androgens and estrogens. It is transformed from its original form into androgens and estrogens in different tissues, such as in skeletal muscle (Vitale, Cesari & Mari 2016). DHEA levels peak at age 20–30 and progressively decrease over time, reaching about 20% of peak levels by age 70–80 (Zjacic-Rotkvic, Kavur & Cigrovski-Berkovic 2010; Ferrari et al. 2001; Heaney, Carroll & Phillips 2013). This hormone displays a constant pattern of secretion and plays a role in cognitive function and in the pathogenesis (origin and development) of several age-related diseases. It is also, in combination with estrogen, one of the key hormones that balance the effects of cortisol.
Both DHEA and cortisol are stress hormones of the HPA axis, but they have opposing effects on the brain and immune system: Cortisol is immunosuppressive and promotes neuronal degeneration, whereas DHEA is immune-enhancing and stimulates neuronal long-term potentiation (i.e., it protects against damage) (Ferrari et al. 2001; Heaney, Carroll & Phillips 2013). Homeostasis is maintained when levels of these two hormones are normal, but age-related declines in DHEA disrupt the cortisol–DHEA balance. Cortisol levels are then higher in relation to DHEA and are no longer counterbalanced as effectively. This skewed ratio has been linked to the development of sarcopenia (Vitale, Cesari & Mari 2016). Unlike cortisol, which follows a diurnal rhythm—high in the morning and low in the evening—DHEA has a flatter pattern of secretion throughout the day (Heaney, Phillips & Carroll 2012).
CORTISOL
Cortisol is released by the HPA axis in response to challenging situations. Unlike other hormones that decline with age, cortisol levels are not affected by the passing of time (Archer et al. 2011; Heaney, Phillips & Carroll 2012; Ferrari et al. 2001). Although cortisol levels appear to be higher in older adults, evidence suggests this is due to the skewed ratio with age-declining hormones, which leads to cortisol being overrepresented. In essence, cortisol’s effect is higher because levels of the hormones that usually balance its effect are now lower. Numerous studies show the correlation between chronic cortisol excess and a variety of negative health outcomes, including
- a neurotoxic effect on the brain;
- a negative influence on cognition, socioemotional functioning, mood, sleep, attention and memory;
- a loss in muscle mass and strength; and
- an increased risk of fracture and osteoporosis in older adults (Ebner et al. 2015; Vitale, Cesari & Mari 2016; Heaney, Phillips & Carroll 2012; Zjacic-Rotkvic, Kavur & Cigrovski-Berkovic 2010).
ESTROGEN
Estrogen may be the most recognized hormone, owing to its key role in one of the most prominent hormonal phases of the female lifecycle: menopause. This is the only endocrine system that undergoes an abrupt and universal age-related change in function. Similar to the change in the cortisol-to-DHEA ratio, the change in the ratio between estrogen and other hormones plays a major role in the function of physiological, neurological and metabolic systems.
With the loss of estrogen, postmenopausal women are left without its antiglucocorticoid and anti-inflammatory responses, as well as its neuroprotective effect on cognitive aging and its role in protecting against muscle mass and strength loss (Vitale, Cesari & Mari 2016; Lowe, Baltgalvis & Greising 2010). Ebner et al. (2015) suggest that higher stress responses in older woman than in older men may be related to estrogen changes and to increased HPA axis responses to psychosocial stress.
TESTOSTERONE
Testosterone, the main anabolic hormone involved in skeletal muscle growth and regeneration, declines steadily with age. The decrease is strongly associated with losses in muscle mass and strength (Lowe, Baltgalvis & Greising 2010; Vitale, Cesari & Mari 2016). Data have shown that testosterone prescriptions increased fivefold between 2000 and 2011 (Wallis 2014; McBride, Carson & Coward 2016), leading to a surge of interest in testosterone replacement therapy. While many factors have contributed to this surge, the increase in the older male population and an “awareness of medical comorbidities” associated with low testosterone, such as metabolic syndrome and cardiovascular disease, appear to be among them.
The decrease in testosterone levels with age can be as much as 0.4%–2% annually after men turn 30 (McBride, Carson & Coward 2016). A study presented at the 2001 annual meeting of The Endocrine Society, however, found that age-related testosterone declines in men may be the result of deteriorating general health, not the cause. In that study, researchers in Australia recruited 325 men over the age of 40 (median age 60) who had “self-reported excellent health and no symptom complaints.” The men provided blood samples nine times over 3 months unless they were taking medication that affected testosterone.
Findings showed that “obesity caused a mild and clinically unimportant lowering of blood testosterone levels and that age had no effect.” While not a conclusive statement on whether or not low levels of testosterone negatively affect aging or vice versa, the findings do shed light on lifestyle habits as a factor in functional decline (The Endocrine Society 2011).
GROWTH HORMONE AND INSULIN-LIKE GROWTH FACTOR TYPE 1
Growth hormone (GH) is secreted by the pituitary gland and stimulates insulin-like growth factor type 1 (IGF-1)—an “extension” of GH—in the liver. GH and IGF-1 work synergistically to prompt muscle growth, although the latter is the primary mediator of muscle repair (Vitale, Cesari & Mari 2016; Laron 2001). Both progressively decline as people age, with daily GH production declining as much as 14% per decade after age 30, and by 50% in men every 7 years (Vitale, Cesari & Mari 2016; Weltman et al. 2006). This downward trend is strongly associated with reduced muscle mass and exercise capacity, exacerbated by the fact that GH is a potential mediator of sarcopenia and an important indicator of an older adult’s functional ability (Kim & Ju 2017).
GH release remains greater in women than in men after age 50, and factors such as age, gender, fitness, nutrition and sleep—as well as regional distribution of body fat—modulate GH secretion (Weltman et al. 2006). One of the most important functions of GH is metabolic and involves stimulating not only lipolysis (lipid breakdown) in adipose tissue but also protein and carbohydrate metabolism (Godfrey & Blazevich 2004). Ottosson et al. (2000), researched the effects of cortisol and GH on body composition and concluded that both act directly on adipose tissue, cortisol being antilipolytic while GH is lipolytic. Interestingly, these researchers found that the effect of GH is strengthened in the presence of cortisol. Combined decreases in testosterone, estrogen and GH levels are known to contribute to an increase in abdominal visceral fat (Weltman et al. 2006).
See also: Here’s to Hormones!
The Role of Exercise in Functional Aging and Hormone Health
There is a vast amount of research on the positive health outcomes that regular physical activity can have in the general population and in older adults, specifically. These outcomes, often related to chronic-disease prevention, improve life expectancy, which, according to the World Health Organization, increased globally by 5.5 years between 2000 and 2016. That’s the fastest increase since the 1960s (WHO 2018). While positive, this poses additional challenges and opportunities for fitness professionals who serve the growing older-adult population.
Physical activity can both influence and be influenced by hormones, depending on the type and intensity of activity, as well as the hormone’s function. According to Godfrey & Blazevich (2004), sleep and exercise are the two most powerful nonpharmacological stimuli of GH secretion. Paterson & Warburton (2010) reported that older adults consider the capacity to carry out everyday activities to be of greater concern than disease prevention. With that in mind, fitness professionals should focus on increasing functional ability and performance, with an ultimate goal of improving quality of life, when working with this niche. The effect that exercise has on hormones can offset some age-related hormonal changes and, therefore, minimize some of the functional decline.
Intensity of Physical Activity
Multiple studies show that exercise intensity is a key factor in the hormone response to physical activity among older adults. Godfrey & Blazevich (2004) found that a significant exercise-induced GH response was achieved with high-intensity training (HIT) and heavy resistance training, but only after a certain amount of time. For example, HIT training above lactate threshold resulted in a GH response only after 10 minutes, and sprints were more effective at 30 seconds versus 6 seconds. This same study showed that, in older adults, acute bouts of heavy resistance training resulted in large-scale and immediate increases in testosterone, DHEA and GH and a decrease in cortisol (Godfrey & Blazevich 2004). Low-intensity exercise, in comparison, had no significant GH response in older adults.
Weltman et al. (2006) also reported that only exercise intensities above lactate threshold elevated GH output in older adults. Similarly, Paterson & Warburton (2010) found that moderate to higher levels of activity, but not low levels, correlated to a 50% reduction in risk of functional limitation and disability. In Kemmler et al. (2003), DHEA-sulfate levels in early postmenopausal females were higher immediately after a combined endurance and strength training session and were still higher 2 hours later. (DHEA becomes DHEA-S in the adrenal glands and the liver.)
Type of Exercise for Functional Aging
Although intensity has the highest effect on hormone output, exercise type and specificity also play crucial roles in functional performance and ability. Kim & Ju (2017) found that both resistance training and aerobic exercise had positive effects on GH and estrogen and that both long-term and short-term aerobic exercise could increase estrogen levels in older adults with very low fitness levels. Testosterone levels, however, appeared to be affected more by resistance training. Lovell et al. (2012) found that testosterone levels increased with 16 weeks of resistance training. Regarding functional outcomes, research has shown that using a combined approach of both aerobic and RT results in more positive outcomes (Paterson & Warburton 2010; Heaney, Carroll & Phillips 2013; Gault & Willems 2013; Kim & Ju 2017).
In contrast to hormone output, functional performance can improve significantly with multiple levels of resistance training. Either heavy or moderate-intensity training can increase functional performance, without a significant difference between the two (Kalapotharakos et al. 2005). The findings in this study suggest that, for optimal neuromuscular adaptations, “heavy RT should be combined with power training or periodized RT in order to emphasize the rapid development of muscle strength that contributes to several daily tasks.”
Exercise specificity is crucial for achieving desired improvements in functional ability. For instance, walking and core instability training increase functional mobility and performance because they engage muscles that are specific to everyday activities, while swimming, although an excellent modality for older adults with bone health issues, is not recommended for increasing functional ability, because specificity is lacking (Paterson & Warburton 2010). Granacher et al. (2013) focused on the importance of the core and concluded that core strength training or Pilates could be used as an alternative to balance and/or resistance training to strengthen trunk muscles and improve spinal mobility—crucial areas of focus when training for optimal performance of everyday activities.
Keep People Moving
The research indicates that, for optimal hormone health and functional ability, older-
adult clients benefit from programs that include both cardiovascular and resistance training, particularly HIT (above lactate threshold), which appears to have the greatest impact. Intensity level is a key factor, with both heavy and moderate-intensity resistance training providing significant functional benefits, while low-intensity does not. That said, it should be noted that walking may greatly reduce one’s disability risk. In fact, walking 4–7 times per week can reduce the onset of disability by 50%–80% in older adults (Paterson & Warburton 2010).
As people age, most of them want to be active and independent and stay as functional as possible. Understanding the role of the underlying mechanisms of functional aging—specifically, the impact of physical activity on hormones—arms fitness professionals to help clients with their active aging goals.
Physical Activity Recommendations for Functional Aging
- Program moderate- to high-intensity exercise for optimal hormone response.
- Provide overload stimulus for all muscles, and keep in mind that eccentric exercises offer additional value, owing to their low energy cost.
- Encourage clients to walk 4–7 times per week (3.3–4.2 METs, 3–4 miles per hour) for reduced risk of disability.
- Recommend a weekly exercise volume goal of 150–180 minutes, with each session being longer than 10 minutes.
- Strive to create programs that last at least 12 weeks. Longer programs are even more successful. For example, 16 weeks of resistance training will increase exercise-induced growth hormone response, and programs lasting more than 24 weeks will significantly increase muscular performance (Godfrey & Blazevich 2004).
Is Age Just a Number?
According to a report issued by the U.S. Department of Health and Human Services (2017), 28.4% of 65–74-year-olds and 44.3% of those 75 and older reported having physical difficulties in 2016. Additionally, 16.9% of the former group and 25.8% of the latter group found it very difficult or impossible to stoop, bend or kneel. Considering these numbers, the emphasis is now turning to improving functional aging rather than just increasing chronological age (USDHHS 2017).
Positive Outcomes With Eccentric Exercises
Research indicates that resistance training is one of the most effective modalities for preserving and improving functional ability in older adults. In exploring variabilities within that modality, Gault & Willems looked at the differences in outcomes for concentric, eccentric and isometric movements. They found that eccentric exercises can be safely used with older adults owing to the muscle’s ability to combine high muscle force with low energy cost. They also concluded that endurance-based, predominantly eccentric exercises involving large muscle groups improved maximal strength and aerobic activity capacity in older adults (Gault & Willems 2013).
References
Archer, T., et al. 2011. Influence of physical exercise on neuroimmunological functioning and health: Aging and stress. Neurotoxicity Research, 20 (1), 69–83.
Dean, W. 2018. Neuroendocrine theory of aging. Accessed Aug. 30, 2018: warddeanmd.com/articles/neuroendocrine-theory-of-aging-chapter-1/.
Ebner, N.C., et al. 2015. Hormones as difference makers in cognitive and socioemotional aging processes. Frontiers in Psychology, 5 (1595).
The Endocrine Society. 2011. Older age does not cause testosterone levels to decline in healthy men. Accessed July 23, 2018: sciencedaily.com/releases/2011/06/110607121129.htm.
Ferrari, E., et al. 2001. Age-related changes of the hypothalamic-pituitary-adrenal axis: Pathophysiological correlates. European Journal of Endocrinology, 144 (4), 319–29.
Gault, M.L., & Willems, M.E.T. 2013. Aging, functional capacity and eccentric exercise training. Aging and Disease, 4 (6), 351–63.
Godfrey, R.J., & Blazevich, A.J. 2004. Exercise and growth hormone in the aging individual, with special reference to the exercise-induced growth hormone response. International Sport Medicine Journal, 5 (4), 246–61.
Granacher, U., et al. 2013. Effects of core instability strength training on trunk muscle strength, spinal mobility, dynamic balance and functional mobility in older adults. Gerontology, 59 (2), 105–13.
Guralnik, J.M. & Melzer, D. 2002. Chronological and functional ageing. In J.R.M. Copeland, M.T. Abou-Saleh, & D.G. Blazer (Eds.), Principles and Practice of Geriatric Psychiatry (2nd ed.). New York: John Wiley & Sons, Ltd.
Heaney, J.L.J., Phillips, A.C., & Carroll, D. 2012. Ageing, physical function, and the diurnal rhythms of cortisol and dehydroepiandrosterone. Psychoneuroendocrinology, 37 (3), 341–49.
Heaney, J.L.J., Carroll, D., & Phillips, A.C. 2013. DHEA, DHEA-S and cortisol responses to acute exercise in older adults in relation to exercise training status and sex. Age, 35 (2), 395–405.
Kalapotharakos, V.I., et al. 2005. Effects of a heavy and a moderate resistance training on functional performance in older adults. Journal of Strength and Conditioning Research, 19 (3), 652–57.
Kim, S., & Ju, S. 2017. Effects of silver gymnastic exercises on aging-related hormones in elderly women living alone. Journal of Physical Therapy Science, 29 (11), 1957–59.
Laron, Z. 2001. Insulin-like growth factor 1 (IGF-1): A growth hormone. Journal of Clinical Pathology: Molecular Pathology, 54 (5), 311–16.
Lovell, D.I., et al. 2012. The hormonal response of older men to sub-maximum aerobic exercise: The effect of training and detraining. Steroids, 77 (5), 413–18.
Lowe, D.A., Baltgalvis, K.A., & Greising, S.M. 2010. Mechanisms behind estrogen’s beneficial effect on muscle strength in females. Exercise and Sport Sciences Reviews, 38 (2), 61–67.
McBride, J.A., Carson, C.C., & Coward, R.M. 2016. Testosterone deficiency in the aging male. Therapeutic Advances in Urology, 8 (1), 47–60.
Nicklas, B.J., & Brinkley, T.E. 2009. Exercise training as a treatment for chronic inflammation in the elderly. Exercise and Sport Sciences Reviews, 37 (4), 165–70.
Ottosson, M., et al. 2000. Effects of cortisol and growth hormone on lipolysis in human adipose tissue. The Journal of Clinical Endocrinology & Metabolism, 85 (2), 799–803.
Paterson, D.H., & Warburton, D.E.R. 2010. Physical activity and functional limitations in older adults: A systematic review related to Canada’s Physical Activity Guidelines. The International Journal of Behavioral Nutrition and Physical Activity, 7 (38).
U.S. Census Bureau. 2017. Older Americans month: May 2017. Profile America Facts for Features, CB17–FF08.
USDHHS (U.S. Department of Health and Human Services). 2017. Health, United States, 2016. Accessed Aug. 31, 2018: cdc.gov/nchs/data/hus/hus16.pdf#042.
Vitale, G., Cesari, M., & Mari, D. 2016. Aging of the endocrine system and its potential impact on sarcopenia. European Journal of Internal Medicine, 35, 10–15.
Wallis, L. 2014. Caveats about testosterone therapy. American Journal of Nursing, 114 (2), 15.
Weltman, A., et al. 2006. Growth hormone response to graded exercise intensities is attenuated and the gender difference abolished in older adults. Journal of Applied Physiology, 100 (5), 1623–29.
WHO (World Health Organization). 2018. Life expectancy. Accessed July 23, 2018: who.int/gho/mortality_burden_disease/life_tables/situation_trends_text/en/.
Zjacic-Rotkvic, V., Kavur, L., & Cigrovski-Berkovic, M. 2010. Hormones and aging. Acta Clinica Croatica, 49 (4), 549.
Maria Luque, PhD, MS, CHES
Maria Luque, PhD, is a health educator, fitness expert, presenter, writer and USAF veteran. She created Fitness in Menopause, a company dedicated to helping women navigate the challenges and rewards of menopause. Her course “Menopausal Fitness: Training the Menopausal Client” is NASM-, AFAA- and ACE- accredited. She holds graduate and postgraduate degrees in health sciences and teaches at the College of Health and Human Services at Trident University International.



