The Art and Science of Intermittent Fasting
This practice may have its roots in ancient tradition, but it is recent research that is bringing to light all the benefits of regularly abstaining from energy intake. Here, learn how to integrate IF into a health-supportive lifestyle.

Intermittent fasting has long been used in ancient traditions and major religions. Recently, it’s grown in popularity within diet culture and health communities. It’s no surprise that a simple Google search of “intermittent fasting” brings up more than 40 million results.
Research reports that more than 40% of American adults are classified as having obesity (Hales et al. 2020; CDC 2020), and 60% have at least one chronic disease (CDC 2019). The health of the masses has become a matter of urgent concern, and, increasingly, researchers are looking to intermittent fasting for its potential to have a positive impact.
Intermittent fasting (IF) can be defined as an eating pattern with periods of little to no energy intake interspersed with periods of normal energy intake, adhered to on a repeating basis. Though scientific evidence on IF is still forming, the latest studies show that IF shows promising benefits for the body and brain. However, eliciting the reported benefits involves more than adopting periods of no energy intake. Just as exercise regimens must be tailored to individuals to be maximally effective, so must the practice of intermittent fasting. The practitioner must be able to commit to IF and integrate it into a holistic program of healthy lifestyle choices designed to enhance overall well-being.
If intermittent fasting has piqued the interest of you or your clients, I invite you to read further to explore the existing research and the implementation of the approach.
Intermittent Fasting: Not Just Another “Diet”
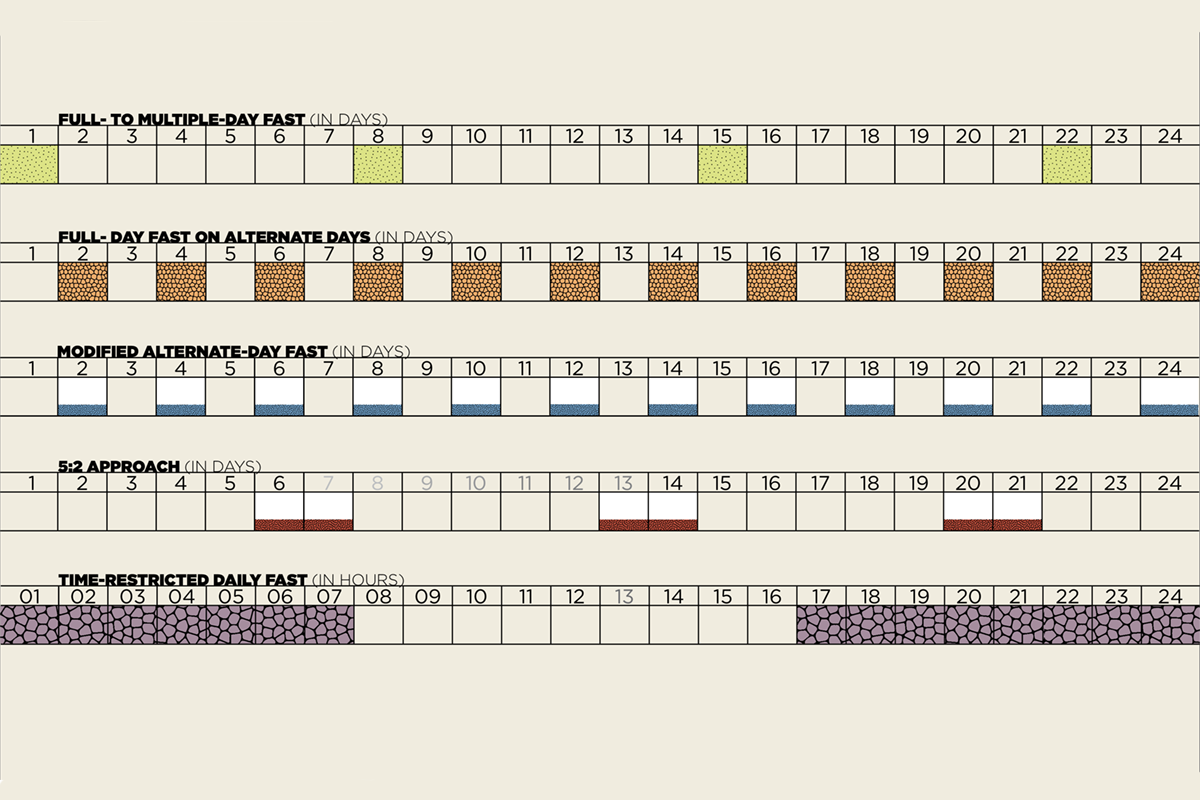
Some intermittent fasting approaches do include caloric restriction but also include food selection, timing and frequency manipulation.
IF has drawn the attention of the weight loss industry because the current approach of caloric restriction continues to show a poor success rate, a high incidence of weight regain and negative biological consequences (Anastasiou, Karfopoulou & Yannakoulia 2015; Ganesan, Habboush & Sultan 2018). When we simply count and restrict calories, it results in physiological and psychological responses that include elevated appetite, reduced energy expenditure, hormonal imbalances, and feelings of guilt and resentment when we fail to sustain a diet’s rigid eating patterns (MacLean et al. 2011).
Further, while research on caloric restriction reports initial improvements in glucose and insulin regulation, physical pain, blood pressure, and energy, these are not prolonged. There’s simply more to weight than an energy-calorie equation. In fact, growing research is showing that meal selection, metabolic processes, circadian rhythms, emotional state and sleep patterns contribute to long-term weight management (Eichelmann et al. 2016; Esser et al. 2014; Kiecolt-Glaser 2010; Leproult & VanCauter 2010; McHill & Wright 2017; Wang et al. 2017). This is where IF differs from dieting.
Some IF approaches do include caloric restriction; however, the method also includes food selection, timing and frequency manipulation. These variables lead to changes in eating and sleeping patterns, resulting in positive alterations to metabolic rhythms (Aksungar et al. 2017). These altered biorhythms, in turn, shift inflammatory biomarkers, hormone secretions, microbiome health, cognitive function and metabolic pathways—all of which can potentially extend life span and, most importantly, lengthen health span (Carlson et al. 2007; Cohen 2018; Longo & Mattson 2014; Mattson, Longo & Harvie 2017).
What IF is not is a quick fix to drop a few pounds. It can take up to 2 months of consistent practice to begin reaping its benefits, with many physiological changes taking closer to a year. That’s why I recommend shifting away from viewing it as a dieting tool and suggest seeing it instead as part of a holistic approach that creates an internal environment of coherency—that is, all systems working together harmoniously. When this happens, we genetically express health and well-being and, in time, weight loss will follow.
See also: Nibbling, Fasting and Feasting: Myths About Meal Timing and Frequency
The Science of Intermittent Fasting: Benefits to Body and Brain
As IF gains momentum in research, the latest scientific findings show promising benefits. A majority of the existing research on IF uses animal models. While some human clinical trials are emerging, not every population has been studied, and the mechanisms behind the reported beneficial outcomes require additional investigation. This does not mean the current findings should be dismissed, as this is how scientific inquiry evolves. We are simply at the beginning of the exploration. What we are finding is that a regular pattern of reduced/no-energy intake sets in motion physiological and neurological shifts that have the potential to address chronic illness and influence the overall aging process. Here’s a review of the key findings:
A Drop in Inflammatory Biomarkers
Chronic inflammation plays a central role in heart disease, cancer, neurological disorders, diabetes, autoimmune illnesses and obesity. Studies have found that, in addition to reducing total cholesterol, LDL levels and triglycerides, IF intervention decreases a number of inflammatory biomarkers, including serum tumor necrosis factor-alpha, C-reactive protein, interleukin-1 and interleukin-6 (Heilbronn et al. 2005; Johnson et al. 2007; Mattson & Wan 2005; Mattson, Longo & Harvie 2017; Varady & Hellerstein 2007). Because of IF’s profound effect on systemic inflammation, the majority of reported benefits will circle back to these biomarkers. This is a delicate dance, as inflammation plays an important role in our immune defenses—but when left unchecked, inflammation sets off an alarm that can lead to hypertension, sugar imbalances, digestive dysfunction, weight struggles and asthma (Mosey 2019).
Positive Changes in Adipose Tissue
Fat tissue is more than a reservoir for stored energy. It acts as an endocrine organ that regulates inflammation, appetite and energy metabolism, and it coordinates with the central nervous system to orchestrate biological processes (Coelho, Oliveira & Fernandes 2013; Kershaw & Flier 2004).
Studies are showing that IF results in an increase in lipolysis for energy needs, leading to weight loss, reduced fat mass, improved leptin and insulin sensitivities, and elevated levels of adiponectin (Gotthardt et al. 2016; Heilbronn et al. 2005; Hoddy et al. 2016; Varady & Hellerstein 2007; Varady et al. 2010; Wan et al. 2010). Leptin and insulin are both regulators of glucose metabolism, and when cell receptivity to these hormones improves, it is possible to prevent or reverse type 2 diabetes (Mattson, Longo & Harvie 2017). Adiponectin, an anti-inflammatory cytokine located in adipose tissue, has been found to have a protective effect against diabetes, heart disease and autoimmune illness (Geagea et al. 2018).
An Enhanced Parasympathetic Nervous System
The parasympathetic nervous system plays a role in the body’s recovery from a fight-or-flight stress response by stimulating cardiorespiratory, digestive and immune activity. IF research is reporting enhanced activation of the PNS with improved intestinal motility, decreased blood pressure and lower resting heart rates (Wan et al. 2014). With heart disease continuing to be the leading cause of death in America, IF’s alterations to PNS activation—with concomitant reductions in inflammatory markers associated with heart disease—makes it a potential strategy recommendation for improving cardiovascular health (Harvie & Howell 2017; Harvie et al. 2013; Johnson et al. 2007; Kroeger et al. 2012; Mattson, Longo & Harvie 2017; Moro et al. 2016).
Improvements in Cognitive Function
Neuroscientists speculate that Alzheimer’s disease and other degenerative brain diseases begin decades prior to noticeable symptoms. This makes our current lifestyle choices central to our future cognition. IF research is showing improved function in key areas of the brain, including the hippocampus, striatum, hypothalamus and brain stem (Longo & Mattson 2014). Short-term to multiple-year studies on IF have shown improvements in verbal memory, global cognition and executive function (Horie et al. 2016; Leclerc et al. 2020; Witte et al. 2009). Research suggests that these beneficial effects are the result of IF-induced increases in brain-derived neurotrophic factor. BDNF is a protein that plays a role in brain development and plasticity, which includes the growth and survival of new neurons, the strengthening of existing neural circuits, and the integration of new neural circuits throughout our life span (Froy & Miskin 2010; Mattson & Wan 2005; Mattson et al. 2018; Wan et al. 2014).
The Art of Intermittent Fasting: Taking an Individualized Approach
Before implementing IF, there are factors to consider. First, is it suitable for the individual in question? As with any food practice, IF is not necessarily appropriate for everyone. For instance, if individuals are looking to counteract poor eating habits or struggling with a chronic sleep disorder, this is not for them. If they’re seeking to enhance a preexisting health-supportive lifestyle, this may be a good choice.
If the answer is yes, the second step is choosing a regimen. So the next factor to consider is, What approach can the individual commit to and consistently do? Options include alternate-day fasting, modified fasting approaches and time-restricted feeding, as described in “A Menu of Intermittent Fasting Regimens,” right. Studies show dropout rates are higher for full-day fasting approaches and compliance is greater for modified or time-restricted options. However, all regimens are initially challenging, as they disrupt daily routines.
They also initiate a stress response in the body, which includes increasing antioxidant defenses, enhancing mitochondrial health, repairing DNA and stimulating autophagy (recycling damaged molecules). IF thereby shifts the body into a repair-and-maintenance state, promoting long-term health (de Cabo & Mattson 2019; Mattson, Longo & Harvie 2017).
That’s the science behind the practice. Reality is there will be a period of physical discomfort, moodiness and preoccupation with food that can last up to a couple of months. This is completely normal. Think about your exercise routine. During recovery, the body adapts to the physiological stress of the exercise session with the potential for “discomfort” in the form of delayed-onset muscle soreness. It’s similar when beginning IF: When you apply an acute stress (fasting), it is natural to experience some imbalance or discomfort as the body adapts.
Aligning Intermittent Fasting With Biological Rhythms
Everything about our being is interconnected, which is why IF is recommended for those with a preexisting health-supportive lifestyle. No action alone creates health. Every action affects all of you.
As IF research expands, connections within the body’s holistic network continue to surface. It’s already known that our digestion, immunity and brain are intricately enmeshed (Mosey 2019). Additional findings show how our microbiome, circadian rhythms and sleep are also part of this web of function (Mosey 2020). As we explore different IF regimens, their relationship to the rest of the body, including its internal rhythms, comes into the conversation. Two types of rhythm that are important for successful IF implementation are the migrating motor complex and the circadian rhythms.
IF and the Migrating Motor Complex
The migrating motor complex is a cyclic electrical pattern in the digestive tract that occurs after the digestion of a meal. Referred to as the gut’s “housekeeper,” MMC is responsible for clearing away residual food and cellular debris and controlling the overgrowth of bacteria in the intestines (Deloose & Tack 2016). This is a four-phased, 90-minute process that begins after a meal has been digested, indicating that it takes about 5 hours between meals to complete the cycle. Every bite we take—snack or meal, healthy or processed—shuts this process off and sends it back to the beginning. Research demonstrates that MMC activity is altered in those who have systemic inflammation and are struggling with obesity (Takahashi 2013).
As much as it has been ingrained in us to “eat three meals a day plus snacks,” this pattern has no scientific basis and does not elevate metabolism, boost immunity, improve glucose levels or increase calorie burn, as is touted in diet-industry marketing. Eating so frequently actually inhibits pathways that help us deal with oxidative and metabolic stress—for example, by recycling damaged molecules and regulating inflammation. Studies show that these pathways are suppressed in those who overeat, are sedentary and do not activate fasting mechanisms (de Cabo & Mattson 2019; Mattson & Arumugam 2018).
We’re also learning that when we do fast—allowing the MMC to complete its process—we become metabolically flexible. Periods of fasting lead to metabolic switching, where the body shifts from liver-derived glucose to adipose tissue ketones as its primary source of energy. (Ketones are more than fuel. They act as signaling molecules affecting the activity of molecules that influence health and aging.) Being able to flip the metabolic switch and maintain cellular functioning creates metabolic flexibility. Those who overeat cannot effectively switch between glucose and fat oxidation, resulting in insulin resistance, reduced leptin sensitivity and obesity (Harvie & Howell 2017; Muoio 2014).
Ultimately, we’re designed to eat, then fast—not graze all day. I know this brings on a sense of panic, as I’ve witnessed it in my own clients’ eyes while I’ve been explaining this aspect of digestion. I assure clients they are not going to starve, become nutrient-deficient or faint from hunger.
Can people feel hungry and moody when they initially begin IF? Of course. Our feeding times are on a cycle, based on how we choose to eat—keyword choose. There’s a mindset that comes with our eating patterns. It’s never just about the food itself, and the practices associated with IF are going to bring those beliefs to the surface to be challenged. IF is also going to bring awareness to physical versus emotional hunger and invite a healthier relationship with food: one where intuition and mindfulness can reside (Mosey 2019).
As mentioned earlier, with any lifestyle change, health-supportive or not, there will be a time of transition as the body undergoes hormonal and metabolic adjustments. When this happens, remember that we’re designed to go 24 hours without food and, in fact, this is quite beneficial to our overall well-being. The good news: Research shows that reductions in initial depression, anger, fatigue and tension—along with improvements in vigor and body-image perception—begin after only a few days to weeks of IF intervention (Harvie et al. 2011; Harvie et al. 2013; Hoddy et al. 2015).
IF and Circadian Rhythms
Along with MMC, IF is linked to circadian rhythms, often known as our internal clock. This roughly 24-hour cycle plays a role in appetite, metabolic rate, hormone secretions, emotional state and sleep patterns (Froy & Miskin 2010; Patterson et al. 2015; Patterson & Sears 2017).
In addition to what we eat, when we eat matters. Our circadian rhythm regulates glucose and lipid oxidation. Depending on time of day, these aspects of energy metabolism are stimulated or downregulated, with the highest insulin sensitivity and thermic effect of food occurring in the morning (Poggiogalle, Jamshed & Peterson 2018). Gastric emptying and blood flow are greater during the day, while metabolic responses to glucose are slower in the evening (Sanders & Moore 1992; Takahashi et al. 2018). Metabolic hormones such as insulin, glucagon, corticosterone, leptin and ghrelin are all regulated through the circadian clock (Sutton et al. 2018).
In recent research, when IF was aligned with circadian rhythms—eating earlier in the day, with dinner prior to 4 p.m.—hunger decreased and there were improvements in insulin sensitivity, blood pressure, fat mass loss, lipid profiles and inflammatory markers (Gill & Panda 2015; Moro et al. 2016; Sutton et al. 2018). When participants ate in the evening, however, they either experienced none of these changes or showed worse metrics (Carlson et al. 2007; Stote et al. 2007; Takahashi et al. 2018).
In sum, the most up-to-date findings suggest that IF with full MMC completion and in alignment with circadian rhythms may be the most beneficial form of IF practice. Keep in mind that it takes a minimum of 12 hours of no energy intake for metabolic blood concentrations to initiate fasting benefits (Cahill 1970; Patel et al. 2019).
See also: Intermittent Fasting: A Primer
Enhancing Intermittent Fasting With a Health-Supportive Environment

Creating a health-supportive internal environment involves sleep, microbiome health and exercise.
To recap: The art of practicing IF requires choosing an appropriate regimen, spacing meals to allow the MMC to complete its process and aligning meal timing with circadian rhythms. Next comes creating a health-supportive internal environment. Three lifestyle factors linked to a beneficial IF regimen are sleep, microbiome health and exercise.
IF and Sleep
Sleep is a metabolically active time when brain and body functions are being renewed and upgraded. During sleep we consolidate memories, regulate emotions and inspire creativity (Vandekerckhove & Wang 2018). It’s also a time to boost immunity, balance hormones, regulate metabolism and repair muscles/tissues. When we are sleep-deprived, these physiological processes become compromised, contributing to the onset of heart disease, obesity, diabetes, cancer and depression (McHill & Wright 2017).
Studies show that physiological responses to sleeping and fasting have some similarities. These include elevated gluconeogenesis, mitochondrial biogenesis, increased lipid catabolism and elevated leptin secretion (Patterson et al. 2015). There is also a bidirectional relationship between circadian rhythms and sleep: IF supports the expression of circadian rhythms, while restful sleep improves the internal clock’s regulation of appetite, metabolism and long-term weight.
IF and the Gut Microbiome
The gut microbiome has its own internal rhythm, which is synchronized with the master circadian clock. These two rhythms collectively regulate digestion, immunity, neural chemistry and sleep (Voigt et al. 2016; Wang et al. 2017). The gut microbiome also plays a role in emotional states (Mosey 2020).
The gut microbiome consists of thousands of bacteria strains, with each producing specific neurotransmitters, immune molecules and metabolites. These include serotonin, dopamine and melatonin, all of which influence stages of sleep (Li et al. 2018; Petra et al. 2015). When sleep deprivation disrupts circadian rhythms, the composition of gut microbes is altered, leading to a hormone release that stimulates hunger, creates cravings, slows metabolism and promotes systemic inflammation (Anderson et al. 2017). It’s a cyclical relationship, with sleep and microbiome health influencing one another.
Along with taking steps to get a full night’s sleep, the recommendation is to eat a primarily plant-based diet. Plants contain immune-boosting and microbiome-supporting compounds, both of which are essential to overall health and longevity (Harland & Garton 2016; Mosey 2019; Toumpanakis, Turnbull & Alba-Barba 2018).
See also: Your Gut Microbiome and Its Connection to Sleep
IF and Exercise
Now comes exercise—the cherry on top. Research shows that exercise amplifies the benefits of IF intervention. Recent findings indicate that the benefits of IF in relation to cardiovascular health, energy metabolism and cognitive function are similar to those found with a consistent exercise program (Mattson, Longo & Harvie 2017). The acute stress stimulus of exercise and the cellular stress of IF both activate signaling pathways that create an adaptive cellular resistance response and boost antioxidant defenses, all of which together help to manage the body’s inflammation alarm system and ward off illness. Maintaining an IF regimen combined with regular exercise results in long-term adaptations that improve physical, mental and emotional well-being while decreasing the risk of chronic disease (de Cabo & Mattson 2019).
Is Intermittent Fasting a Good Fit?
As a fitness professional, you can encourage clients to use the following information to decide whether
intermittent fasting may work for them personally, but be sure to stay within your scope of practice.
IF may be a good fit for those who want to
- improve body awareness and hunger/fullness cues;
- explore life without strong food attachments;
- work toward the potential health benefits of the practice; and
- enhance an existing health-supportive lifestyle.
IF is not meant for those who are
- younger than 23 or older than 75;
- pregnant or breastfeeding;
- diagnosed with advanced type 1 diabetes;
- struggling with a chronic sleep disorder;
- challenged with an eating disorder, including orthorexia;
- attempting to justify rigid eating rules;
- fearful of gaining weight;
- trying to counteract poor eating habits;
- focused on food only as related to weight and body image; or predisposed to taking practices, including healthy ones, to an unhealthy extreme.
Getting Started With Intermittent Fasting
Fitness professionals have long known to start a new exercise program slowly and use microprogressions to maintain safety and minimize discomfort. A similar philosophy can be applied to intermittent fasting. With scope of practice and client safety top of mind, here are some thoughts that clients and the nutrition professional you refer them to may want to consider:
- Choose an IF regimen carefully. Select an approach that can be sustained: Consistency is crucial to reaping benefits.
- Change the timing gradually.Rather than shift the timing all at once, trim a little time off the eating window, about 30 minutes, each day.
- Omit late-night eating.This will help align IF with circadian rhythms.
- Time meals to support digestion.Consider going 5 hours between meals or snacks to support the body’s migrating motor complex and create metabolic flexibility.
- Stay hydrated.Drink plenty of filtered water. Keep herbal teas to a minimum and avoid caffeinated beverages.
- Rely primarily on plants.To improve potential benefits, eat a health-supportive, mostly plant-based diet.
- Expect some discomfort.There will be a time of imbalance as hunger sensations and hormones adapt. Be prepared to feel hungry, moody and preoccupied with food when first starting an IF regimen.
- Listen to your body.After your body has adapted, continue to pay attention to its responses to fasting. Do not force yourself to fast.
- Be committed.This is not a one-time experiment. Practice for at least 8 weeks to begin reaping IF’s benefits as part of a preexisting healthy lifestyle.
- Be flexible. IF creates physiological changes and psychological shifts. It also alters socialization around food. Remember: It’s a practice requiring overall consistency, not perfection.
Some Final Words on Intermittent Fasting
What we do, day in and day out—our dominant thoughts, emotions and actions—is what determines our overall well-being. A health-supportive lifestyle is one where there’s balance between action and rest, doing and being. It’s one that attends to a healthy diet, physical exercise, mindfulness and restorative sleep. Whether IF becomes a part of that is a personal choice. As I tell my clients, there are many pathways toward finding wellness. Simply find what resonates with you and be consistent in your actions.
References
Aksungar, F.B., et al. 2017. Comparison of intermittent fasting versus caloric restriction in obese subjects: A two year follow-up. The Journal of Nutrition, Health and Aging, 21 (6), 681–85.
Anastasiou, C.A., Karfopoulou, E., & Yannakoulia, M. 2015. Weight regaining: From statistics and behaviors to physiology and metabolism. Metabolism, 64 (11), 1395–407.
Anderson, J.R, et al. 2017. A preliminary examination of gut microbiota, sleep, and cognitive flexibility in healthy older adults. Sleep Medicine, 38, 104–107.
Cahill, G.F. 1970. Starvation in man. The New England Journal of Medicine, 282 (12), 668–75.
Carlson, O., et al. 2007. Impact of reduced meal frequency without caloric restriction on glucose regulation in healthy, normal weight middle-aged men and women. Metabolism, 56 (12), 1729–34.
CDC (Centers for Disease Control and Prevention). 2019. Chronic diseases in America. Accessed Apr. 21, 2020: cdc.gov/chronicdisease/resources/infographic/chronic-diseases.htm.
CDC. 2020. Adult obesity facts. Accessed Apr. 21, 2020: cdc.gov/obesity/data/adult.html.
Coelho, M., Oliveira, T., & Fernandes, R. 2013. Biochemistry of adipose tissue: An endocrine organ. Archives of Medical Science, 9 (2), 191–200.
Cohen, L.J. 2018. Fasting the microbiome to treat diabetes. Science Translational Medicine, 10 (441), eaat8529.
de Cabo, R., & Mattson, M.P. 2019. Effects of intermittent fasting on health, aging, and disease. The New England Journal of Medicine, 381 (26), 2541–51.
Deloose, E., & Tack, J. 2016. Redefining the functional roles of the gastrointestinal migrating motor complex and motilin in small bacterial overgrowth and hunger signaling. American Journal of Physiology: Gastrointestinal and Liver Physiology, 310 (4), G228–33.
Eichelmann, F., et al. 2016. Effect of plant-based diets on obesity-related inflammatory profiles: A systematic review and meta-analysis of intervention trials. Obesity Reviews, 17 (11), 1067–79.
Esser, N., et al. 2014. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Research and Clinical Practice, 105 (2), 141–50.
Froy, O., & Miskin, R. 2010. Effect of feeding regimens on circadian rhythms: Implications for aging and longevity. Aging, 2 (1), 7–27.
Ganesan, K., Habboush, Y., & Sultan, S. 2018. Intermittent fasting: The choice for a healthier lifestyle. Cureus, 10 (7), e2947.
Geagea, A.G., et al. 2018. Adiponectin and inflammation in health and disease: An update. Open Medicine Journal, 5, 20–32.
Gill, S., & Panda, S. 2015. A smartphone app reveals erratic diurnal eating patterns in humans that can be modulated for health benefits. Cell Metabolism, 22 (5), 789–98.
Gotthardt, J.D., et al. 2016. Intermittent fasting promotes fat loss with lean mass retention, increased hypothalamic norepinephrine content, and increased neuropeptide Y gene expression in diet-induced obese male mice. Endocrinology, 157 (2), 679–91.
Hales, C.M., et al. 2020. Prevalence of obesity and severe obesity among adults: United states, 2017–2018. NCHS Data Brief No. 360. Hyattsville, MD: National Center for Health Statistics. Accessed May 2, 2020: cdc.gov/nchs/data/databriefs/db360-h.pdf.
Harland, J., & Garton, L. 2016. An update of the evidence relating to plant-based diets and cardiovascular disease, type 2 diabetes and overweight. Nutrition Bulletin, 41 (4), 323–38.
Harvie, M., & Howell, A. 2017. Potential benefits and harms of intermittent energy restriction and intermittent fasting amongst obese, overweight and normal weight subjects—a narrative review of human and animal evidence. Behavioral Sciences, 7 (1), 1–22.
Harvie, M., et al. 2013. The effect of intermittent energy and carbohydrate restriction v. daily energy restriction on weight loss and metabolic disease risk markers in overweight women. The British Journal of Nutrition, 110 (8), 1534–47.
Harvie, M.N., et al. 2011. The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: A randomized trial in young overweight women. International Journal of Obesity, 35 (5), 714–27.
Heilbronn, L.K., et al. 2005. Alternate-day fasting in nonobese subjects: Effects on body weight, body composition, and energy metabolism. The American Journal of Clinical Nutrition, 81 (1), 69–73.
Hoddy, K.K., et al. 2015. Safety of alternate day fasting and effect on disordered eating behaviors. Nutrition Journal, 14 (44).
Hoddy, K.K., et al. 2016. Changes in hunger and fullness in relation to gut peptides before and after 8 weeks of alternate day fasting. Clinical Nutrition, 35 (6), 1380–85.
Horie, N.C., et al. 2016. Cognitive effects of intentional weight loss in elderly obese individuals with mild cognitive impairment. The Journal of Clinical Endocrinology and Metabolism, 101 (3), 1104–12.
Johnson, J.B., et al. 2007. Alternate day calorie restriction improves clinical findings and reduces markers of oxidative stress and inflammation in overweight adults with moderate asthma. Free Radical Biology and Medicine, 42 (5), 665–74.
Kershaw, E.E., & Flier, J.S. 2004. Adipose tissue as an endocrine organ. The Journal of Clinical Endocrinology and Metabolism, 89 (6), 2548–56.
Kiecolt-Glaser, J.K. 2010. Stress, food, and inflammation: Psychoneuroimmunology and nutrition at the cutting edge. Psychosomatic Medicine, 72 (4), 365–69.
Kroeger, C.M., et al. 2012. Improvement in coronary heart disease risk factors during an intermittent fasting/calorie restriction regimen: Relationship to adipokine modulations. Nutrition and Metabolism, 9 (98).
Leclerc, E., et al. 2020. The effect of caloric restriction on working memory in healthy non-obese adults. CNS Spectrums, 25 (1), 2–8.
Leproult, R., & Van Cauter, E. 2010. Role of sleep and sleep loss in hormonal release and metabolism. Endocrine Development, 17, 11–21.
Li, Y., et al. 2018. The role of microbiome in insomnia, circadian disturbance and depression. Frontiers in Psychiatry, 9 (669).
Longo, V.D., & Mattson, M.P. 2014. Fasting: Molecular mechanisms and clinical applications. Cell Metabolism, 19 (2), 181–92.
MacLean, P.S., et al. 2011. Biology’s response to dieting: The impetus for weight regain. American Journal of Physiology: Regulatory, Integrative and Comparative Physiology, 301 (3), R581–600.
Mattson, M.P., & Arumugam, T.V. 2018. Hallmarks of brain aging: Adaptive and pathological modification by metabolic states. Cell Metabolism, 27 (6), 1176–99.
Mattson, M.P., Longo, V.D., & Harvie, M. 2017. Impact of intermittent fasting on health and disease processes. Aging Research Reviews, 39, 46–58.
Mattson, M.P., & Wan, R. 2005. Beneficial effects of intermittent fasting and caloric restriction on the cardiovascular and cerebrovascular systems. The Journal of Nutritional Biochemistry, 16 (3), 129–37.
Mattson, M.P., et al. 2018. Intermittent metabolic switching, neuroplasticity and brain health. Nature Reviews: Neuroscience, 19 (2), 63–80.
McHill, A.W., & Wright, K.P. 2017. Role of sleep and circadian disruption on energy expenditure and in metabolic predisposition to human obesity and metabolic disease. Obesity Reviews, 18 (1, Suppl.), 15–24.
Moro, T., et al. 2016. Effects of eight weeks of time-restricted feeding (16/8) on basal metabolism, maximal strength, body composition, inflammation, and cardiovascular risk factors in resistance-trained males. Journal of Translational Medicine, 14 (290), 1–10.
Mosey, T. 2019. The food-inflammation connection. Fitness Journal, 16 (7), 18–25.
Mosey, T. 2020. The Gut-Sleep Connection. Fitness Journal, 17 (2), 18-27.
Muoio, D.M. 2014. Metabolic inflexibility: When mitochondrial indecision leads to metabolic gridlock. Cell, 159 (6), 1253–62.
Patel, S., et al. 2019. GDF15 provides an endocrine signal of nutritional stress in mice and humans. Cell Metabolism, 29 (3), 707–18.
Patterson, R.E., & Sears, D.D. 2017. Metabolic effects of intermittent fasting. Annual Review of Nutrition, 37, 371–93.
Patterson, R.E., et al. 2015. Intermittent fasting and human metabolic health. Journal of the Academy of Nutrition and Dietetics, 115 (8), 1203–12.
Petra, A.I., et al. 2015. Gut-microbiota-brain axis and its effect on neuropsychiatric disorders with suspected immune dysregulation. Clinical Therapeutics, 37 (5), 984–95.
Poggiogalle, E., Jamshed, H., & Peterson, C.M. 2018. Circadian regulation of glucose, lipid, and energy metabolism in humans. Metabolism, 84, 11–27.
Sanders, S.W., & Moore, J.G. 1992. Gastrointestinal chronopharmacology: Physiology, pharmacology and therapeutic implications. Pharmacology and Therapeutics, 54 (1), 1–15.
Stote, K.S., et al. 2007. A controlled trial of reduced meal frequency without caloric restriction in healthy, normal-weight, middle-aged adults. The American Journal of Clinical Nutrition, 85 (4), 981–88.
Sutton, E.F., et al. 2018. Early time-restricted feeding improves insulin sensitivity, blood pressure, and oxidative stress even without weight loss in men with prediabetes. Cell Metabolism, 27 (6), 1212–21.
Takahashi, M., et al. 2018. Effects of meal timing on postprandial glucose metabolism and blood metabolites in healthy adults. Nutrients, 10 (11), 1763.
Takahashi, T. 2013. Interdigestive migrating motor complex: Its mechanism and clinical importance. Journal of Smooth Muscle Research, 49, 99–111.
Toumpanakis, A., Turnbull, T., & Alba-Barba, I. 2018. Effectiveness of plant-based diets in promoting well-being in the management of type 2 diabetes: A systematic review. BMJ Open Diabetes Research and Care, doi:10.1136/bmjdrc-2018-000534.
Vandekerckhove, M., & Wang, Y. 2018. Emotion, emotion regulation and sleep: An intimate relationship. AIMS Neuroscience, 5 (1), 1–17.
Varady, K.A., & Hellerstein, M.K. 2007. Alternate-day fasting and chronic disease prevention: A review of human and animal trails. The American Journal of Clinical Nutrition, 86 (1), 7–13.
Varady, K.A., et al. 2010. Improvements in body fat distribution and circulating adiponectin by alternate-day fasting versus calorie restriction. The Journal of Nutritional Biochemistry, 21 (3), 188–95.
Voigt, R.M., et al. 2016. Circadian rhythm and the gut microbiome. International Review of Neurobiology, 131, 193–205.
Wan, R., et al. 2010. Cardioprotective effect of intermittent fasting is associated with an elevation of adiponectin levels in rats. The Journal of Nutritional Biochemistry, 21 (5), 413–17.
Wan, R., et al. 2014. Evidence that BDNF regulates heart rate by a mechanism involving increased brainstem parasympathetic neuron excitability. Journal of Neurochemistry, 129 (4), 573–80.
Wang, Y., et al. 2017. The intestinal microbiota regulates body composition through NFIL3 and the circadian clock. Science, 357 (6354), 912–16.
Witte, A.V., et al. 2009. Caloric restriction improves memory in elderly humans. Proceedings of the National Academy of Sciences of the United States of America, 106 (4), 1255–60.
Teri Mosey, PhD
Teri Mosey is the founder of Holistic Pathways and has been working in various arenas of the health and fitness industry since 1996. Her knowledge and experience has developed over the years to include studies of Eastern and Western philosophies, yogic perspectives, energy medicine, meditative practices and culinary exploration. Through personal and group dynamics, Teri provides supportive and resourceful environments to assist others in discovering their personal path to wellness. She presents alternative perspectives of biochemistry, human physiology and nutrition by incorporating the principles of holistic living into her work. Teri believes with a dynamic foundation of knowledge combined with personal responsibility and freedom of choice, we can all recognize the power within us to co-create our health and ultimately our lives.





